Urine Crystals (Crystalluria)
Urine Crystals (Crystalluria)
Sample for urine crystals
- Freshly voided urine is the best sample.
- Refrigerate or if you keep it at room temperature that will increase the formation of the crystals.
Precautions for urine crystals (Crystluria)
- pH is very important to note.
- Taking the history of the medications will save time and energy.
- Refrigeration will precipitate out many crystals, because of the change in the solubility of various crystals.
- Urine kept at room temperature leads to precipitation or dissolves the crystals.
- The radiographic dye can make crystals in dehydrated patients.
- Ampicillin and sulfonamides also give rise to crystal formation, this happens in dehydrated patients.
Definition of urine crystals (Crystlluria)
- There may be well-defined crystals or amorphous material in the urine sediment.
- These appear as geometrical formed structures or amorphous materials.
- The presence of crystals in the urine is called Crystalluria.
- When urine is left at room temperature or refrigerated then urine becomes cloudy because of the precipitation of crystals or amorphous material.
Importance of the urine crystals (crystalluria):
- These crystals are important in the case of kidney stones.
- Renal damage was caused by the crystals.
- In liver diseases.
- Inborn error of metabolism.
- Some of the crystals indicate some metabolic disorders like cystinuria or a sulfa drug.
- The stone formation may be without crystals in the urine or crystalluria may be without stone formation.
- Crystals are seen mostly in concentrated urine.
- Crystals are divided into :
- Normal or abnormal
- In alkaline or acidic urine.
- Crystals were found due to medication.
- Crystals found in acidic urine have a pH of <6.5, and in alkaline urine, pH is >7.0.
Reporting of the crystalluria:
- Rare/HPF.
- FeW/HPF.
- Moderate/HPF.
- Many/HPF.
Mechanism of the formation of urine crystals:
- Kidneys are the main site for the excretion of the waste product of metabolism, which is needed by the body.
- Urea is from the metabolism of the amino acid.
- Creatinine from the muscles.
- Uric acid from the nucleic acid.
- The hemoglobin end product is bilirubin.
- Hormones are excreted as hormone metabolites.
- Kidneys also excrete toxins and other foreign substances produced by the body or ingested, such as pesticides, drugs, and food additives.
Process of crystals formation:
- Crystals are formed by the crystalization of urine solutes.
- The solutes are:
- Inorganic salts.
- Organic compounds.
- Medications by drug use.
- Precipitation is dependent upon:
- Temperature.
- pH.
- Solute concentration.
- As the concentration of solutes increases, their ability to remain in solution decreases and results in crystal formation.
- Solute precipitates more readily at low temperatures.
- So if you keep the urine at room temperature or refrigerate it, then crystals are abundant.
- Organic and iatrogenic compounds crystalize more easily in the acid urine.
- Inorganic salts are less soluble in neutral or alkaline pH.
- The exception is calcium oxalate which precipitates in both acidic and alkaline mediums.
- The Slower crystalization leads to larger crystal formation, but the basic structure remains unchanged.
- Reversal of the pH will dissolve the crystals.
- Amorphous urates crystals formed in the refrigerated sample; if you warm the urine, these will disappear.
- Amorphous phosphate crystals need acetic acid to dissolve. But practically, this is not done because the acetic acid will distort the RBCs.
Helping point in Reporting the crystals:
- Always note the pH of the urine, which will help to identify the crystals.
- All abnormal crystals are found in acidic urine.
- Polarized microscopy also helps to identify the crystals.
Characteristics of crystals:
- Most of the urate crystals are yellow to reddish-brown.
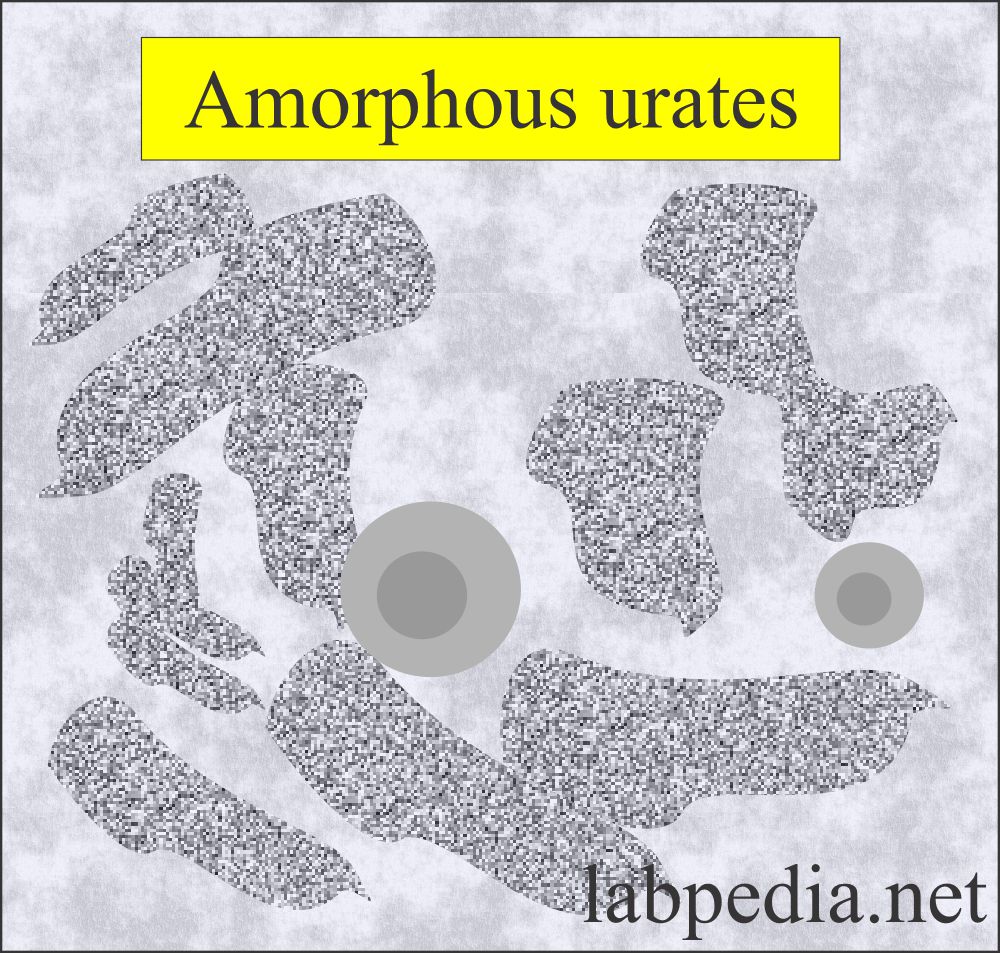
- Amorphous urates appear in yellow-brown granules. These are in clumps and look like the granular cast.
- If urine is refrigerated then the amorphous urates crystals precipitate and give pink sediment.
- Amorphous urates crystals appear in the urine with low pH of >5.5, and uric acid crystals appear when the pH is lower.
Various types of crystals in acid urine:
Acidic- urine crystals are:
- Uric acid.
- Amorphous urates.
- Sodium urates.
- Cystine (these are rarely found).
- Cholesterol crystals (these are rarely found).
- Tyrosine (these are rarely found).
- Leucine (these are rarely found).
- Bilirubin.
Uric acid:
- Serum uric acid is raised in 40% to 50% of the patients.
- These are seen in various shapes like four-sided flat plates, wedge-shaped, and rosettes.
- These are usually yellow-brown in color.
- But maybe colorless and six-sided shapes like cystine crystals.
- These are birefringent in polarized light.
- These are common in patients with leukemia getting chemotherapy.
- Sometimes these are seen in gout.
Acid urates and sodium urates:
- These are like amorphous urates and are seen in less acidic urine.
- These are seen along with amorphous urates and have little clinical significance.
- Sodium urate crystals are needle-shaped and are seen in the synovial fluid during the attack of gout and also appear in the urine.
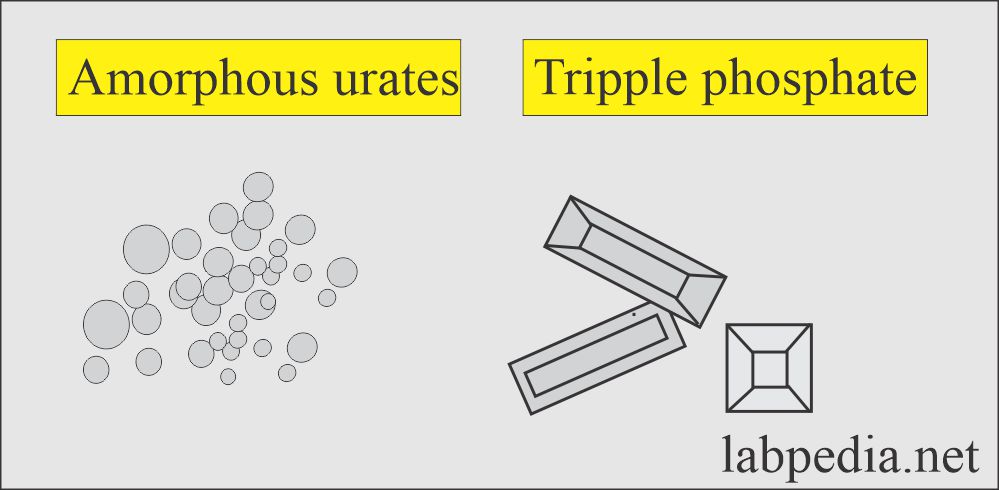
Amorphous urates:
- These are yellow-brown granules.
- These may appear in clumps resembling granular cast.
- These are brick dust or yellow-brown.
- These crystals are found in acidic urine with a pH >5.5 (acidic urine).
- These are soluble in an alkaline medium.
Calcium oxalate:
- More common in acidic urine but may be seen in neutral or alkaline urine.
- The most common is dihydrate, which is a colorless, octahedral envelope shape or two pyramids joined together at their bases.
- Monohydrate crystals are oval or dumble-shaped.
- In polarized light, both are birefringent.
- The finding of clumps of calcium oxalate crystals indicates renal stone formation.
- These are commonly seen when taking foods like tomato, asparagus, and ascorbic acid.
- Monohydrate crystals are seen in ethylene glycol (antifreeze) poisoning.
- These are soluble in dilute hydrochloric acid.
Crystals in the acidic urine :
| Name of crystals | pH | Effect of heating (solubility) | Shape | Color |
|
acidic (<acidic) | soluble at 60 C | amorphous | Colorless to yellow, Amorphous, or large granules |
|
acidic <5.5 | alkali-soluble | rhombic, four-sided flat plates | yellow-brown |
|
acidic >5.5 | alkali and heat | amorphous or sand-like | Microscopically yellow-brown and occurs in clumps |
|
Acidic | |||
|
acidic or alkaline | dilute HCL | enveloped shaped, dumbbell-shaped | The colorless octahedral envelope or two pyramids joined at the base. |
|
acidic | Dilute HCl acid | hexagonal | Colorless |
|
acidic | Chloroform | rectangular, notched plates | Colorless |
|
acid/neutral | Alkali or heat | needles shape form clumps or rosettes | Colorless to yellow, needles |
|
acidic /neutral | Hot alkali or alcohol | spheres with a concentric circle or radial striations | yellow-brown |
|
acid |
Acetic acid, HCL, ether or chloroform |
clumped needles or granular | yellow color |
|
acid/neutral | Acetone | Rosette form, needle, | colorless to yellow-brown |
|
acid/neutral | in needles form | colorless | |
|
acid | 10% NaOH | like cholesterol | colorless |
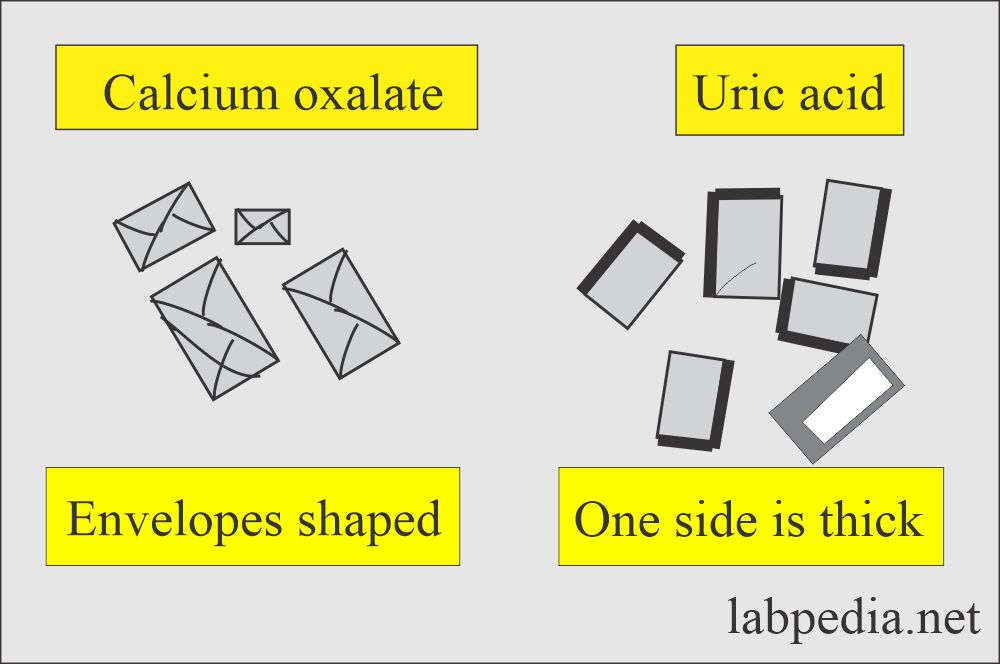
Urine Crystals (Crystalluria): Calcium oxalate and uric acid crystals
Various types of crystals in alkaline urine:
Alkaline urine crystals are:
- Calcium phosphate.
- Amorphous phosphates.
- Calcium carbonate.
- Ammonium biurate.
- Tripple phosphate.
Amorphous phosphate:
- These are granular in shape, like amorphous urates.
- If refrigerated, then these produce a white precipitate, which does not dissolve on warming.
- These are differentiated from the urates by color and the pH of the urine.
- These are soluble in an alkaline or neutral medium.
- These are soluble in dilute acetic acid.
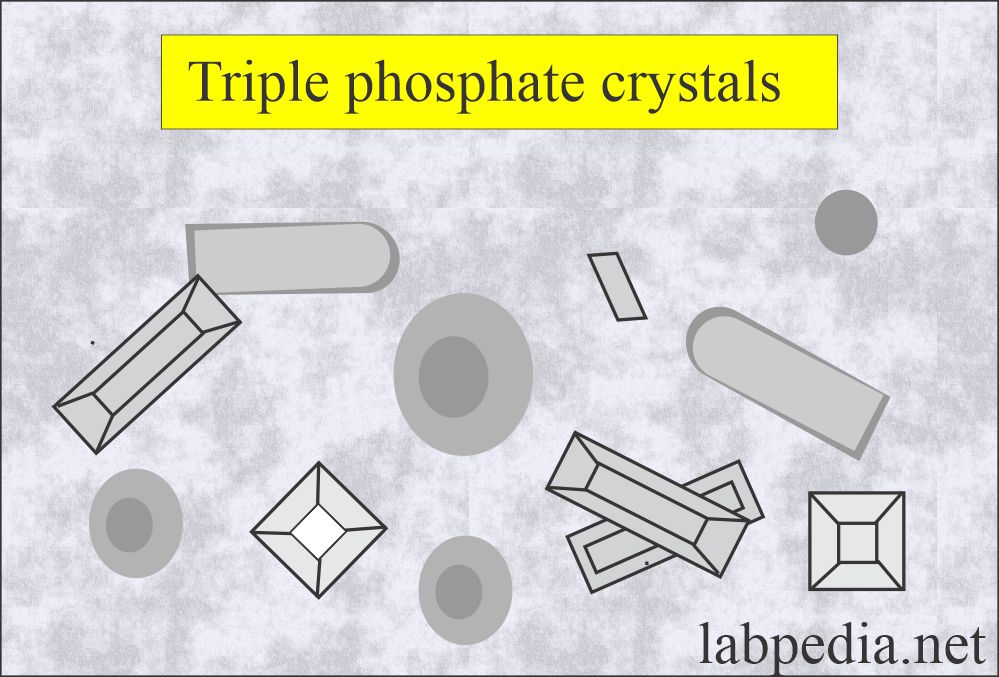
Triple phosphate (Ammonium magnesium phosphate):
- These are seen in alkaline urine.
- These are colorless, prism-shaped, resembling the coffin lid.
- Under polarized lights are birefringent.
- These have no clinical significance.
- These are seen in an alkaline medium.
- These are soluble in dilute acetic acid.
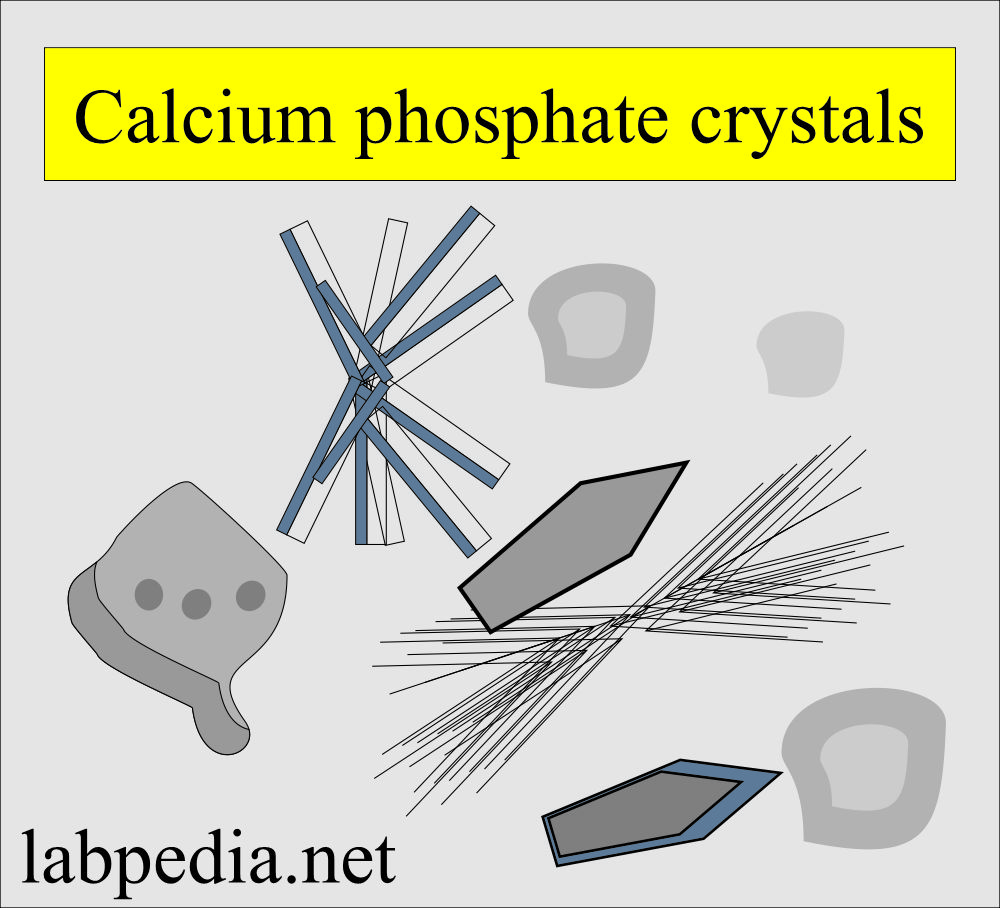
Calcium phosphate:
- These are colorless, flat rectangular plates.
- Or thin prism and often in rosette forms.
- Rosette forms need to be differentiated from the sulphonamides crystals.
- These crystals dissolve in dilute acetic acid while sulphonamides crystals will not.
- These have no clinical significance.
- These are seen inpH of alkaline or neutral.
- These are soluble in dilute acetic acid.
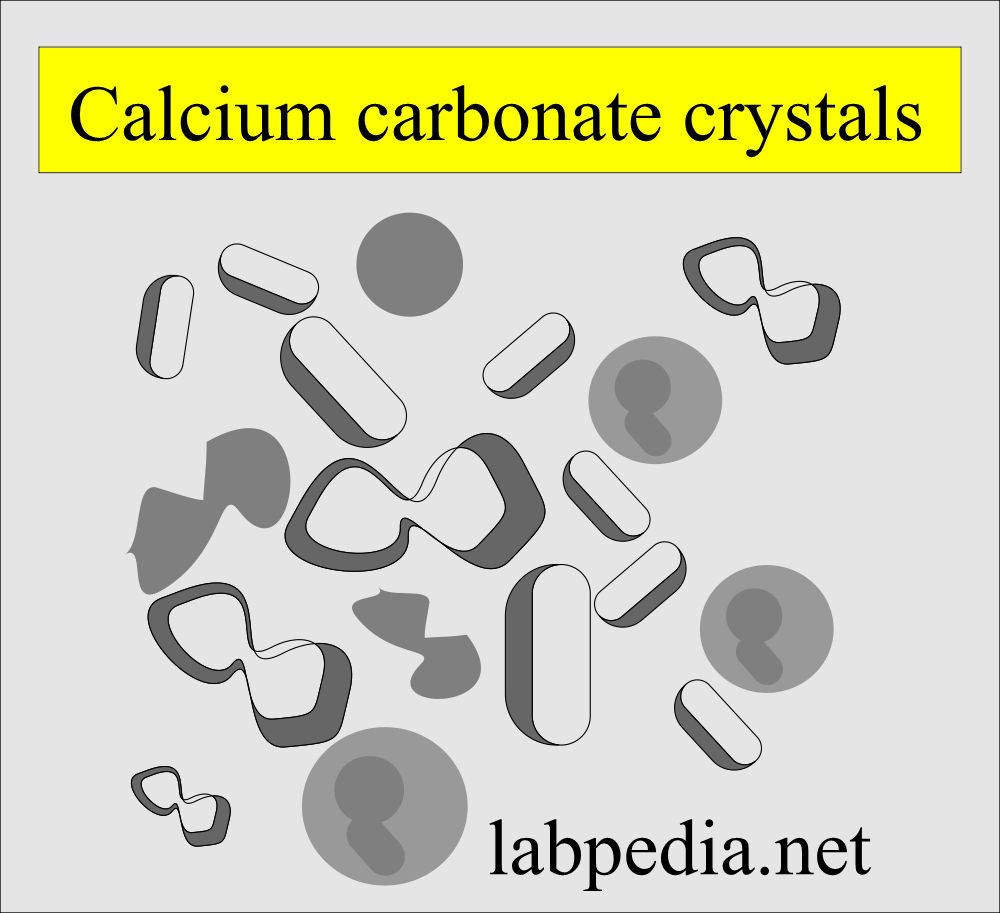
Calcium carbonate:
- These are small. colorless, with a dumbbell or spherical shapes.
- These may occur in clumps and resemble amorphous material.
- If you add acetic acid, then there is gas formation.
- In polarized light are birefringent and this differentiates from the bacteria.
- Thes crystal has no clinical significance.
- These crystals are seen in an alkaline medium.
- These are soluble in acetic acid with gas formation.
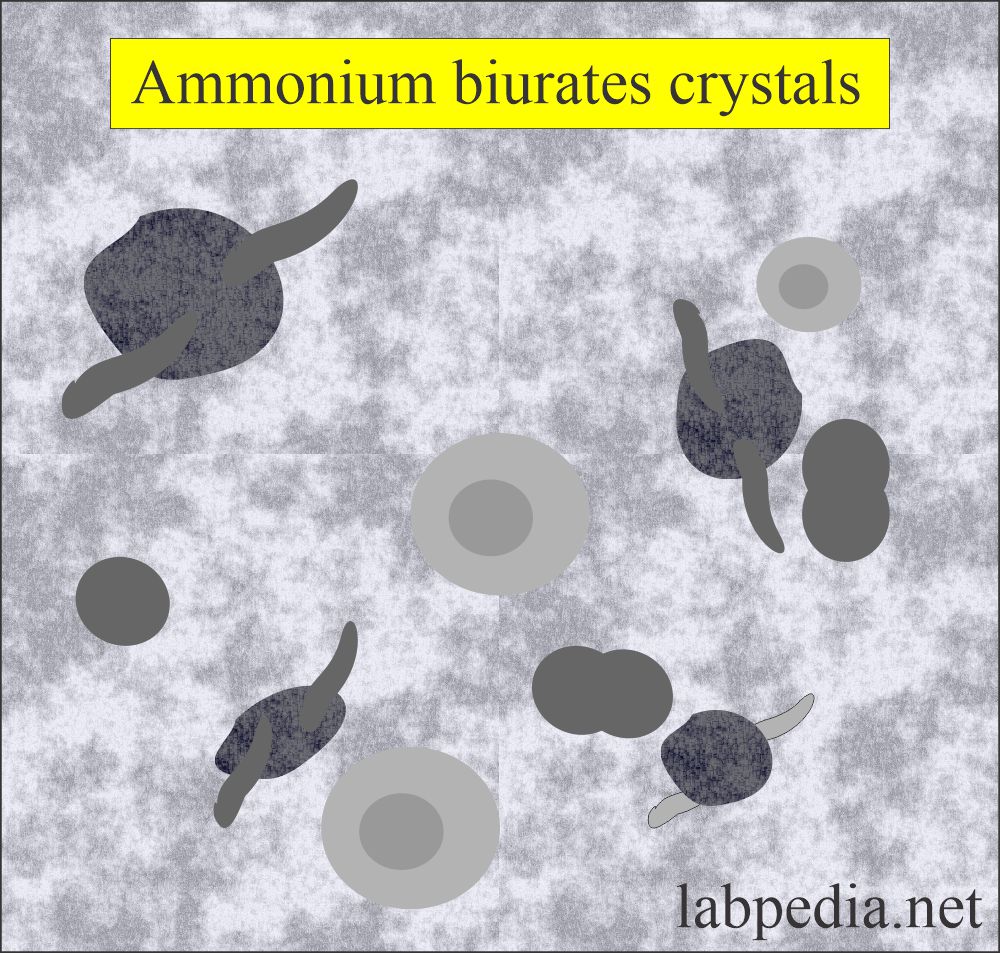
Ammonium biurates:
- These have characteristic yellow-brown colors.
- These are usually described as thorny apples, because of the spicule-covered spheres.
- These dissolve at 60 °C.
- If you add glacial acetic acid, then these will change into uric acid.
Crystals in alkaline urine :
| Name of crystals | pH | Effect of heat | Color and shape |
|
Alkaline | Remain insoluble | Granular |
|
Alkaline | Colorless and prism-shaped like a coffin lid | |
|
Alkaline | Remain insoluble | Colorless, flat rectangular plates or thin prisms often in rosettes |
|
Alkaline | Small, colorless with a dumbbell or spherical shape, it may occur in clumps and resemble amorphous material. | |
|
Alkaline | Dissolve at 60 °C | Yellow-brown color, thorny apples |
The solubility of crystals:
| Crystals | Color | pH | Soluble in |
|
Colorless | Acid/Neutral | Dilute HCl acid |
|
Yellow-brown | Acid | Alkali |
|
Brick dust or yellow-brown | Acid | Alkali and when heated |
|
Colorless | Alkaline | Dilute acidic acid |
|
White-colorless | Alkaline/neutral | Dilute acid acid |
|
Colorless | Alkaline/neutral | Dilute acidic acid |
|
Colorless | Alkaline | Acetic acid |
|
Yellow-brown | Alkaline | Acetic acid with heat |
Abnormal crystals:
- Metabolic origin:
- Tyrosine.
- Cystine.
- Cholesterol.
- Leucine.
- Bilirubin.
- Hemosiderin.
- Drugs origin:
- Sulfonamides.
- Radiographic contrast media.
- Ampicillin.
Cystine crystals:
- These are seen in the inborn error metabolic disorder when the cystine is not absorbed by the renal tubules (cystinuria).
- There is a tendency to form renal calculi.
- These are colorless, hexagonal plates and may be thick or thin.
- In the presence of ammonia, there is the disintegration of these crystals.
- Cysteine stones are seen in 1% to 2% of the cases.
- The cyanide-nitroprusside test is needed to confirm the cystine crystals.
- These crystals appear in an acidic medium.
- It is soluble in ammonia and dilutes HCL.
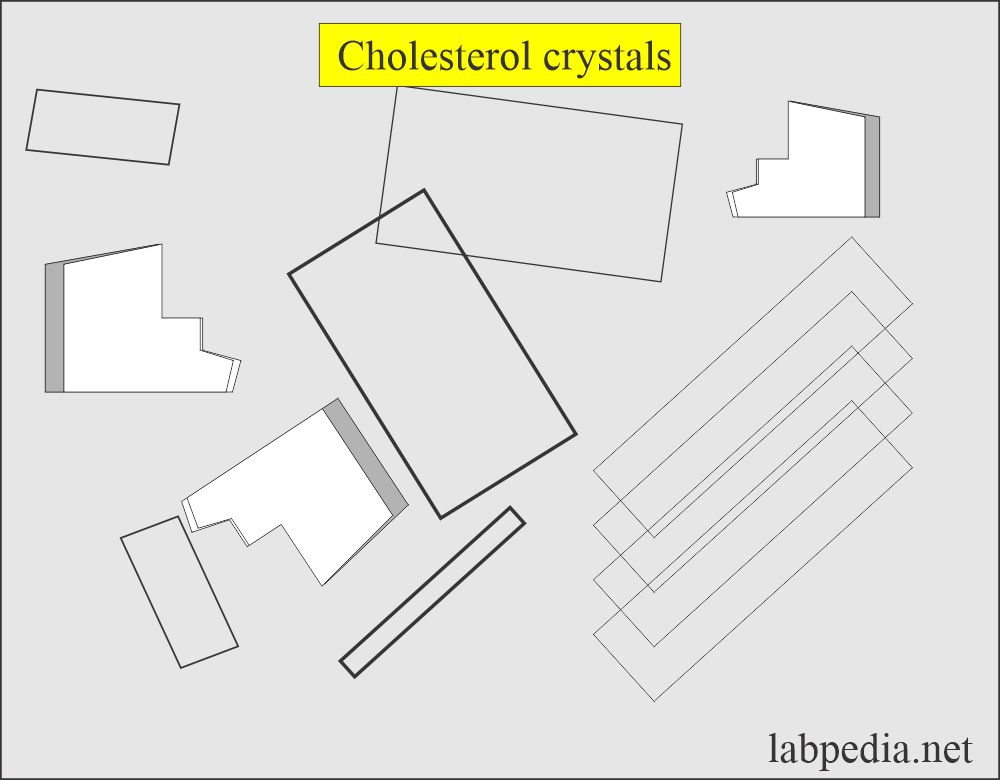
Cholesterol crystals:
- These are not seen unless the urine is refrigerated.
- These have a characteristic appearance resembling rectangular plates with a notch in one or more corners.
- These are seen as disorders producing lipiduria in nephrotic syndrome, along with fatty acids and fat oval bodies.
- In polarized light, these are birefringent.
- This crystal appears in an acidic medium.
- These are soluble in chloroform medium.
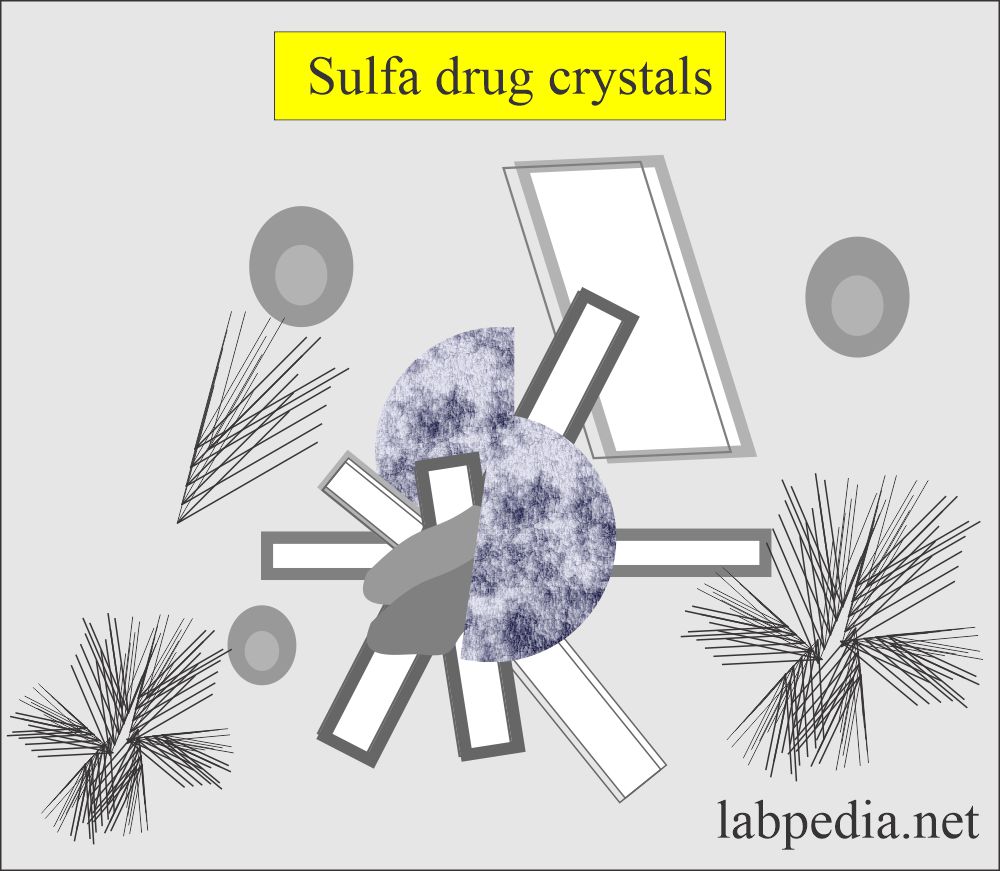
Sulfonamide crystals:
- There are a variety of crystal shapes and colors.
- Shapes variables like needles, whetstone, rhombic, wheat, and rosette with color ranging colorless to yellow-brown,
- The history of the patient will help you to diagnose these crystals.
- Diazo reaction can confirm these crystals.
- These crystals are seen in an acid/neutral medium.
- These crystals are soluble in acetone.
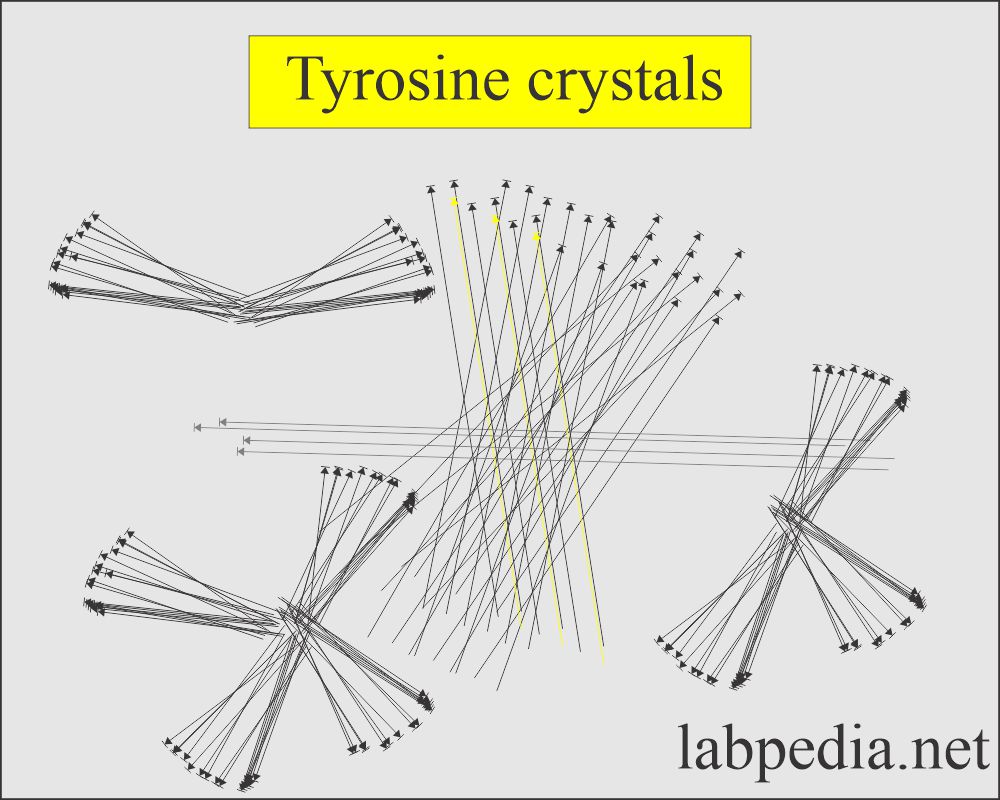
Tyrosine crystal:
- These are fine colorless to the yellow needle-like structures which form clumps or rosettes.
- These may be seen in inherited disorders of amino acid metabolism.
- These crystals are formed in an acid/neutral medium.
- These crystals are soluble in alkali or by heat.
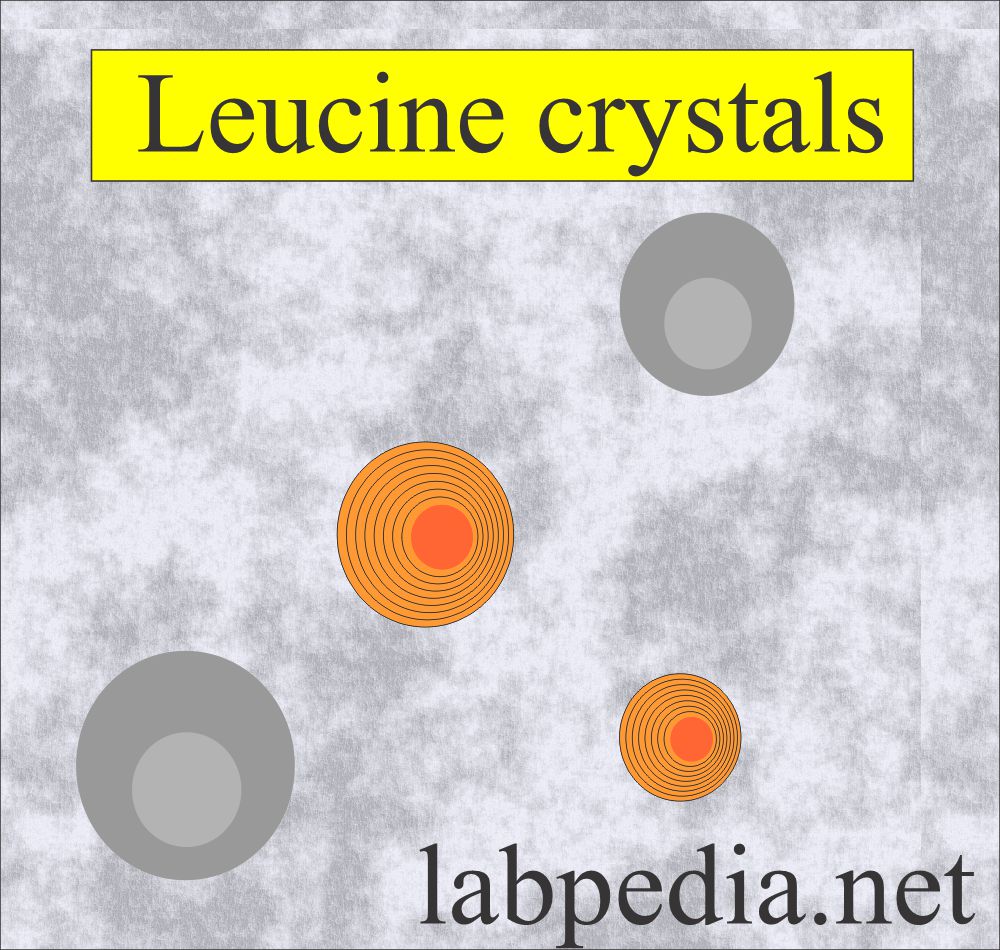
Leucine crystals:
- These are seen because of the defect in the amino acid leucine.
- These are yellow-brown spheres that will show concentric circles and radial striations.
- These are also called wagon wheels.
- These crystals form in an acid/neutral medium.
- These are soluble in hot alcohol and alkali.
- These are less frequent than tyrosine crystals.
- These are accompanied by tyrosine crystals.
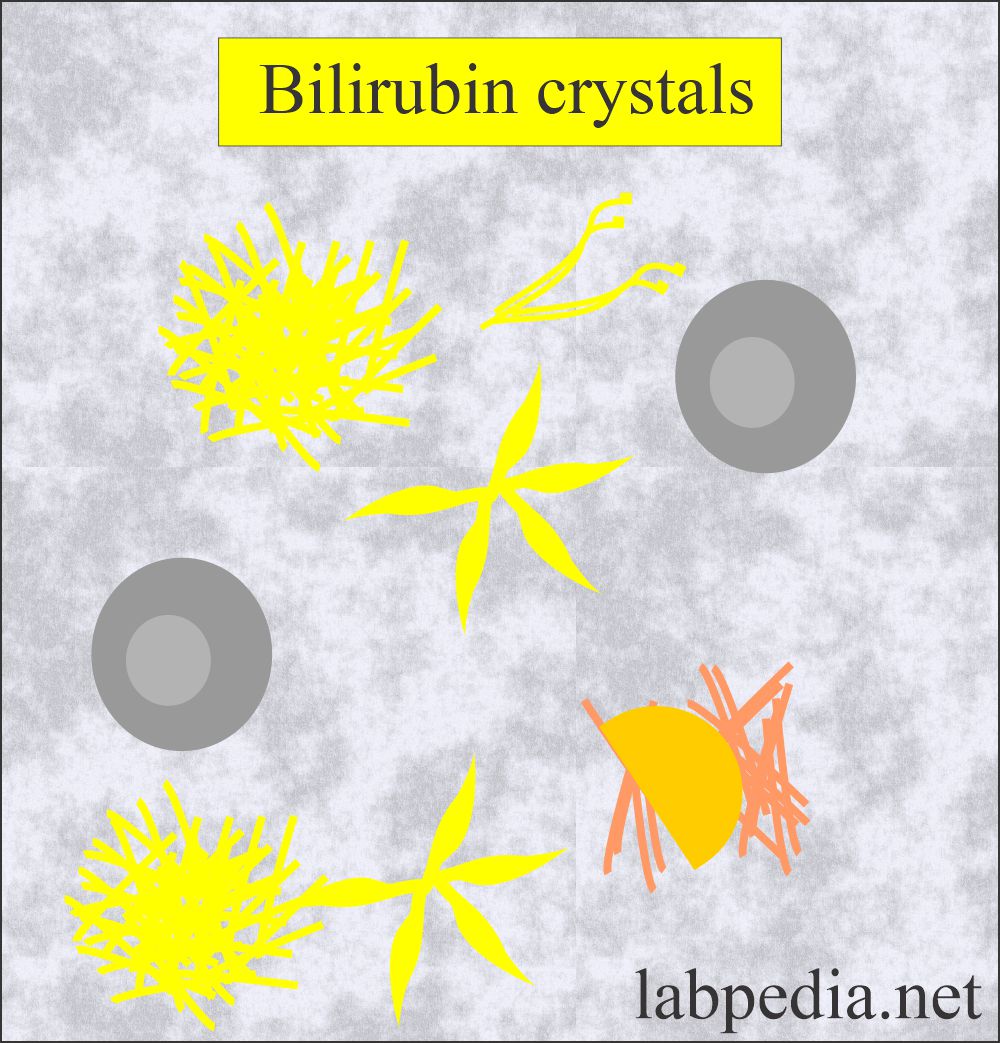
Bilirubin Crystals:
- Bilirubin crystals are present in liver diseases where there is an increased amount of bilirubin excreted in the urine.
- These crystals are clumped needles or granules with the characteristic color of the bilirubin as yellow.
- The chemical reaction for the bilirubin is positive.
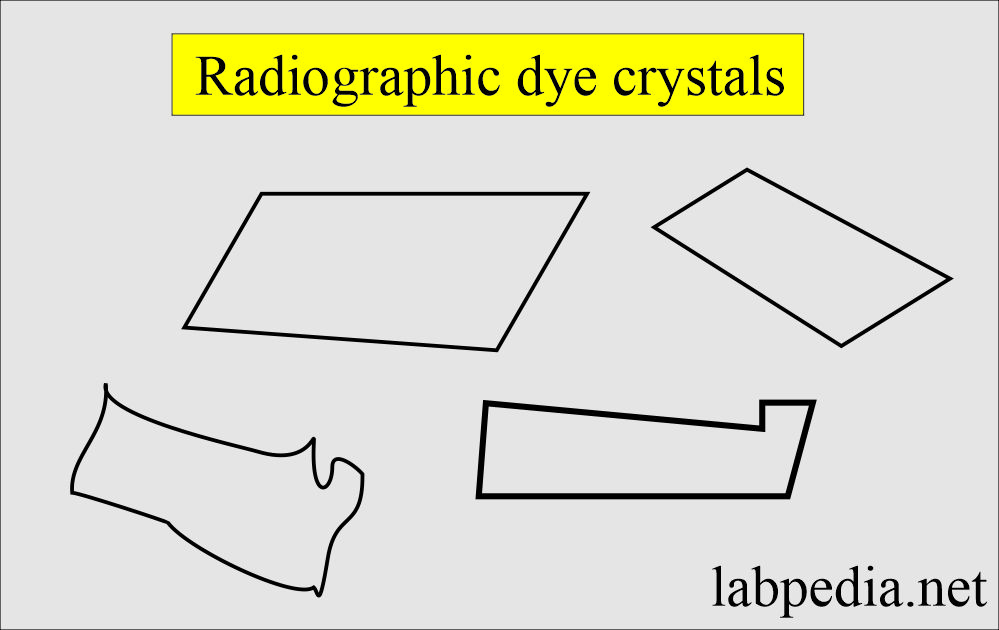
Radiographic material crystals:
- These crystals are colorless.
- These crystals appear in an acid medium.
- These crystals are soluble in 10% NaOH.
Significance of crystals:
- Calcium oxalate crystals in clumps indicate renal stone formation.
- Calcium oxalate crystals are abundant in food with high oxalic acids, like tomatoes, asparagus, and ascorbic acid.
- Monohydrate oxalate crystals are seen in ethylene glycol poisoning (antifreeze material).
Contamination and artifacts:
There are a few contaminants that interfere with the microscopy of the sediments:
- Starch.
- Fibers, including diaper fibers.
- Oil droplets.
- Air bubbles.
- Pollin grains.
- Fecal contamination.
- Glass fragments.
Questions and answers:
Question 1: What are the most common crystals and renal calculi.
Question :
Question 2: What are the artifacts in the urine examination?
Question 3: What will be the pH for the bilirubin crystals??