Iron Metabolism, Total Iron Binding Capacity (TIBC), Transferrin and Transferrin Saturation
Iron Metabolism:
What sample is needed for iron estimation?
- It is mainly the blood that prepares the serum.
How will you define iron?
- Iron is the most abundant element on Earth.
- There is a trace amount in the living cells.
- Most of the iron in humans is located in the porphyrin ring of heme.
- Porphyrins are incorporated into:
- Hemoglobin.
- Myoglobin.
- Cytochrome.
- Peroxidase.
- Catalase.
- There are iron-sulfur proteins like NADH dehydrogenase and succinate dehydrogenase.
What are the functions of iron?
- Iron can interact reversibly with oxygen.
- This action can function in an electron-transfer reaction.
- The above function makes iron biologically indispensable.
- Iron takes part in the transport of the oxygen.
- It takes part in cellular energy generation.
- It takes part in the DNA metabolism.
- Iron has a role in the immune process.
How will you discuss Iron metabolism?
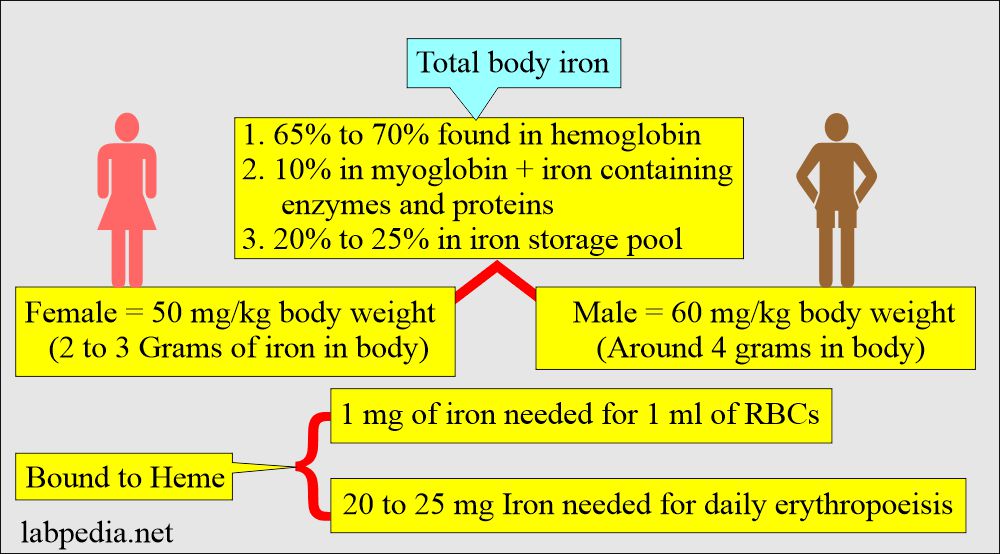
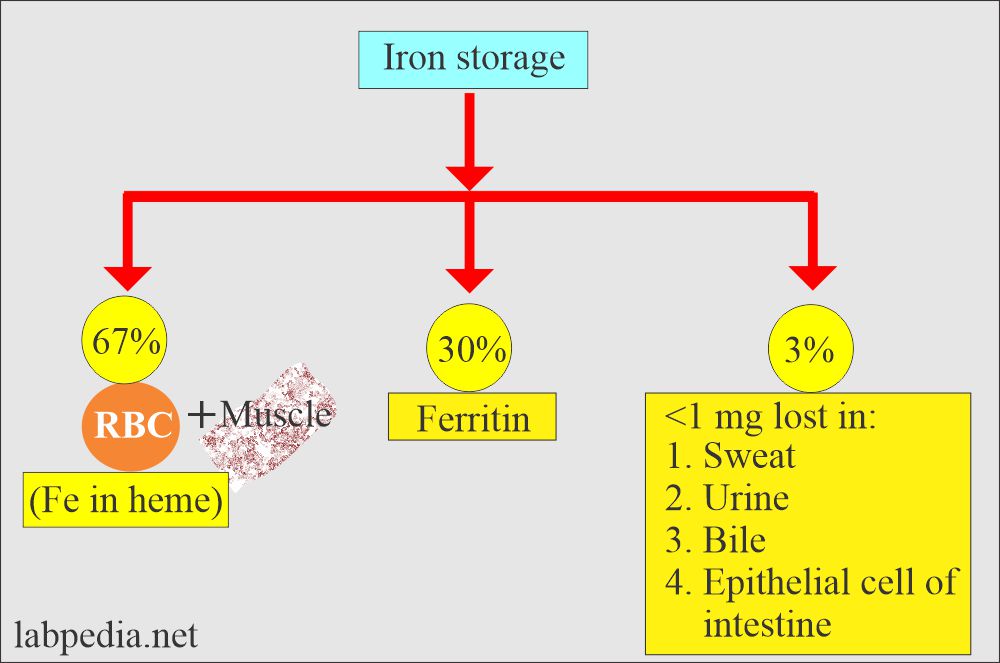
- It is the most abundant trace element in the body.
- 65% is bound to Heme.
- Iron concentration measurement refers specifically to the Fe +++ bound to the transferrin, not to the iron circulating as free hemoglobin in the blood.
- Iron is constituents of:
- Heme.
- Hemoglobin.
- Methemoglobin.
- Myoglobin.
- Several enzymes.
How will you discuss the Iron uptake?
- Iron intake depends on a person’s age, sex, and body requirements.
- Iron intake of 1 mg/day is sufficient for men and postmenopausal women.
- Women during menses lose 20 to 40 mg of iron/per cycle, so they need 2 mg of iron per day.
- During pregnancy, fetal demands, and subsequent breastfeeding, a total loss of 900 mg of iron occurs.
- Iron demand during pregnancy and lactation reaches 3 to 4 mg/day.
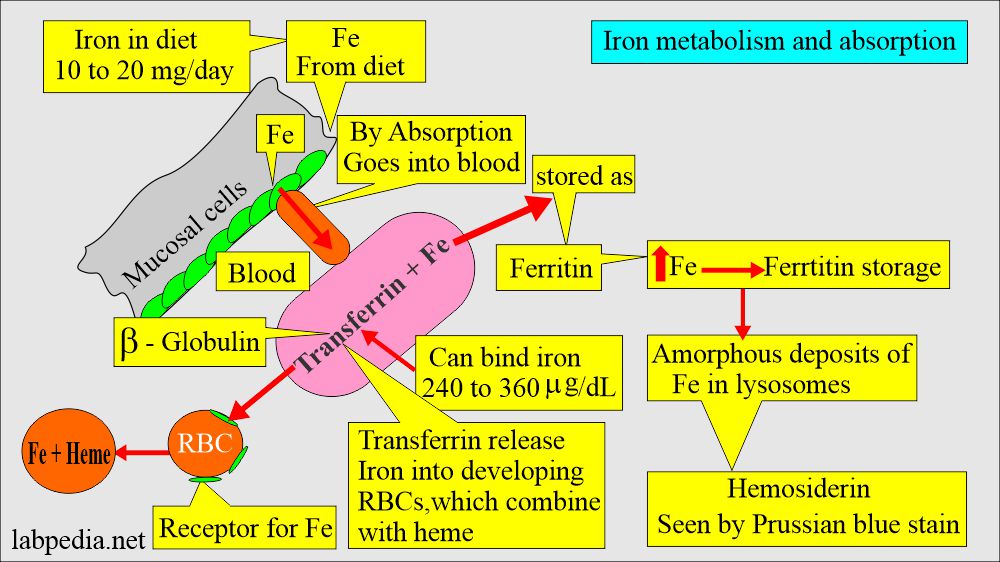
How will you discuss Iron absorption?
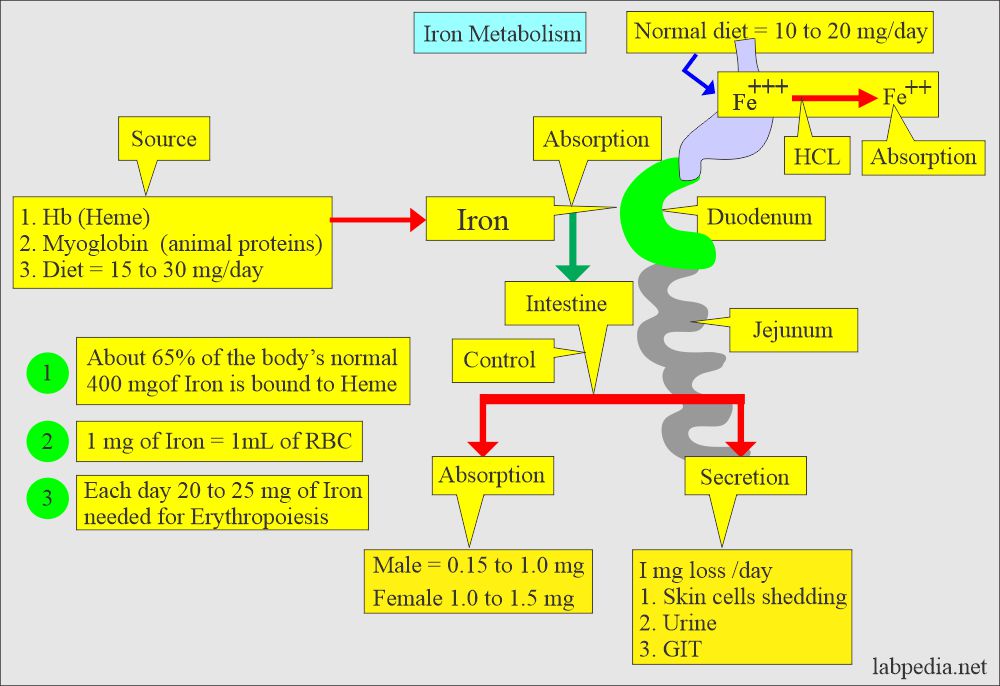
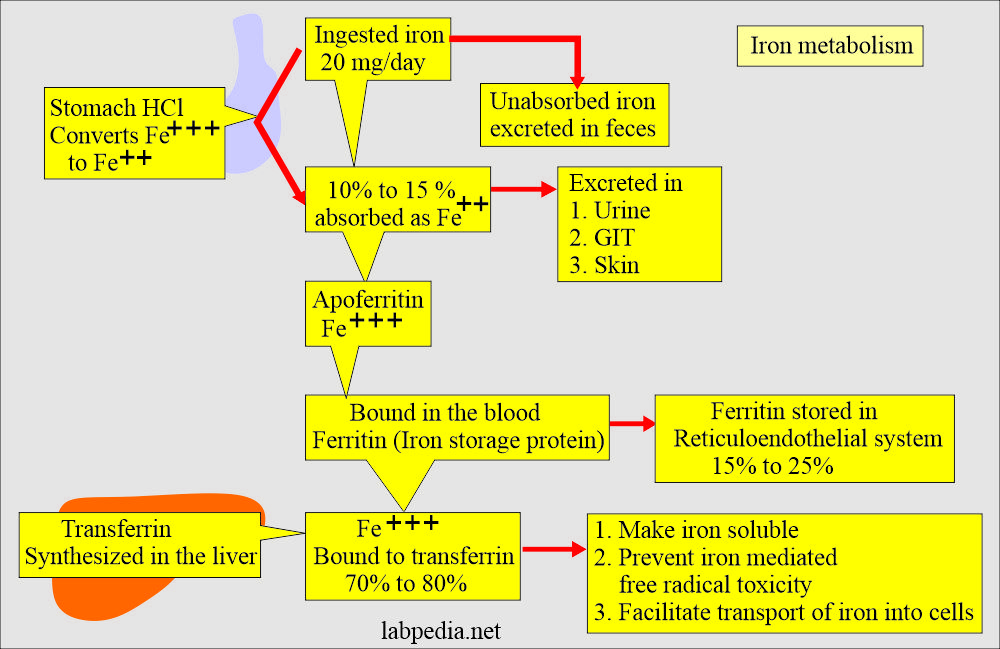
- A normal diet contains 10 to 20 mg of iron per day (the amount varies between references).
- Only 5% to 10% of this amount is absorbed mainly in the duodenum and upper small intestine.
- The intestinal epithelium absorbs iron.
- Iron is ingested in Fe+++ form and is converted to Fe++ form for absorption.
- The conversion of Fe+++ to Fe ++ form takes place in the stomach, where gastric acid HCL provides the acidity to reduce the iron.
- A Ferriruductase enzyme on the brush border of the epithelial cells also reduces Fe +++ to Fe ++ form.
- This Fe++ form is transported into the cell by a divalent metal transport (DMT) system.
What are the factors for Iron absorption?
- Ascorbic acid (vitamin C), sugars, and amino acids increase absorption.
- Phosphates in eggs, cheese, and milk, oxalates and phytates in vegetables, and tannates in tea decrease iron absorption.
- Milk and antacids bind to iron and reduce its absorption.
- Vitamin reduces Ferric (Fe+++) iron to Ferrous (Fe++) by vitamin C, and the ferrous form is absorbed very easily.
What are the sources of Iron loss?
- Iron 1 mg/day is lost in the urine, sweat, bile, and shedding of skin epithelial cells.
- This absorbed iron attaches to the blood’s transport protein (Transferrin).
- So, transferrin may indirectly represent the TIBC.
Total Iron Binding Capacity (TIBC)
What sample is needed for Total Iron Binding Capacity (TIBC)?
- Take blood (3 to 5 mL) to prepare the serum.
- The early morning sample is preferred because of the diurnal variation in the iron concentration.
- Take the sample in the morning (around 10 am) because the iron level varies daily.
- The specimen may be collected as serum without anticoagulant.
- Can use plasma with heparin.
- Reject the hemolysed sample.
What are the Precautions for Total Iron Binding Capacity (TIBC)?
- Do not use anticoagulants like oxalate, citrate, or EDTA because these bind to iron and are unacceptable.
- Twelve hours of fasting is preferred.
- Water intake is allowed.
- Stop taking iron-containing supplements before 24 to 48 hours.
- Chloramphenicol and oral contraceptives can raise the TIBC value.
- Adrenocorticotrophic hormones can produce false negative results.
- If you handle blood samples roughly, it leads to invalidated results.
- Avoid vitamin C, which increases iron absorption. Foods rich in vitamin C include oranges, grapefruit, cabbage, and potatoes.
What are the Indications for Total Iron Binding Capacity (TIBC)?
- This is done to diagnose the anemias. It helps in the differential diagnosis of anemia.
- Other tests needed are Total iron and Transferrin.
- This helps in iron metabolism.
- It is used to screen for iron overload.
- TIBC evaluates:
- Amount of iron.
- Iron storage.
- Nutritional status in anemia.
How will you define Total iron binding capacity (TIBC)?
- TIBC is calculated as follows:
- Transferrin (mg/L) x 0.025 = µmol/L.
- TIBC measures the maximum amount of iron bound to protein and transports iron in the blood.
- The maximum amount is transferred as transferrin.
- It is the capacity of transferrin to bind to iron.
- TIBC is an indirect measurement of Transferrin concentration.
- TIBC measures the total amount of iron that apotransferrin has the capacity to bind.
What is the pathophysiology of Total Iron Binding Capacity (TIBC)?
- TIBC refers to the amount of iron that could be bound by saturation of transferrin and other minor iron-binding proteins in the serum or plasma.
- TIBC is the sum of all proteins bound to iron.
- TIBC increases by 70% in patients with iron deficiency anemia.
- When serum iron falls, then TIBC increases.
- TIBC is increased in the presence of iron deficiency but may be normal or low in chronic diseases.
- TIBC may be calculated from the direct measurement of serum transferrin by the following formula:
- TIBC µg/dL = serum transferrin mg/dL x 1.2521 ( Another reference = Transferrin mg/dL x 0.025).
- A small proportion of the iron is bound to other proteins, so the above equation underestimates the TIBC.
- The amount of apotransferrin still available to bind the iron can be measured in the unsaturated iron-binding capacity.
-
- It is measured as = TIBC minus serum iron (µg/dL).
-
Ferritin:
What sample is needed for the Ferritin level?
- Venous blood is needed to prepare the serum.
What are the indications for Ferritin level?
- It differentiates and classifies anemias.
- It measures the amount of iron storage protein.
- It diagnoses iron deficiency or excess.
- It predicts and monitors iron deficiency.
- It tells the response to iron therapy.
- It differentiates iron deficiency from chronic diseases as a cause of anemia.
- It monitors iron status in patients with chronic renal diseases with or without dialysis.
- It can be used as a population study for iron levels and response to the iron supplement.
What are the precautions for Ferritin?
- Record the age of the patient. Old people have less Ferritin level.
- It is lower in the menstruating women than in the premenopausal age.
How will you define Ferritin?
- It is the chief iron-storage protein in the body.
- It represents stored iron.
How will you interpret Ferritin?
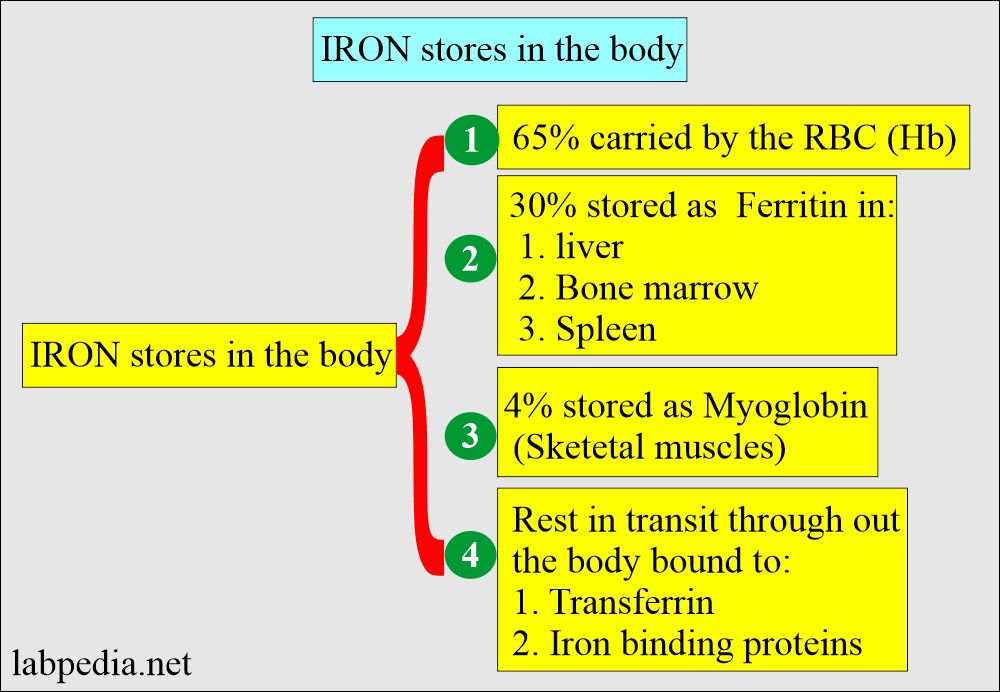
- 10% to 20% – 30% of the total iron is stored as Ferritin.
- Iron is stored as ferritin in the body in the liver, spleen, and bone marrow.
- Or stored as Ferric (Fe +++) bound to an apoferritin protein molecule.
- When iron is needed, it is released from the Ferritin and bound to the β1 globulin molecule, transferrin.
- Serum Ferritin is the best diagnostic test for iron deficiency anemia.
- Because ferritin is the measure of the iron stored in the body.
- In iron deficiency anemia, the ferritin level is <15 µg/L as compared to the normal level of 20 to 250 µg/L.
- In children <, 6 µg/L compared to the normal value of 7 to140 µg/L.
- In baby <, 12 µg/L compared to the normal values of 50 to 200 µg/L.
- Ferritin is the acute phase protein, so its value may increase in infections, SLE, liver diseases, malignancies, and chronic renal failure.
Transferrin:
What sample is needed for Transferrin estimation?
- Collect venous blood to prepare the serum.
What are the indications for Transferrin?
- Transferrin measures iron absorbed from diet sources.
- Transferrin measures iron produced by the breakdown of RBCs.
What are the precautions for Transferrin estimation?
- With estrogen and oral contraceptive therapy, Transferrin levels increase.
- Transferrin level decreases with dextran, corticotropin, corticosteroids, testosterone, and asparaginase.
- Hemolysis may affect the test result.
- Foods like liver, egg yolk, beef, and prune juices are rich in iron.
- Avoid vitamin C-rich foods like grapefruits, oranges, potatoes, and cabbage.
How will you define transferrin?
- Transferrin is a glycoprotein and is responsible for the transport of iron.
- Transferrin transport circulating Fe+++ molecule.
- Normally, only 1/3 of the sites of iron bindings are occupied.
- The remaining unoccupied sites are called unsaturated iron-binding capacity.
- Transferrin transports iron throughout the body and carries it to the cells.
- A low level of transferrin will lead to inadequate formation of hemoglobin and results in anemia.
How will you interpret the Transferrin?
- It represents the major protein that binds to iron. The majority of the iron is bound to transferrin.
- Transferrin is a beta-globulin (β1-globulin).
- Transferrin’s capacity to bind iron in normal plasma is 240 to 360 µg/dL.
- Transferrin also acts as an acute-phase protein.
- This is a transport protein synthesized in the liver.
- This regulates iron absorption.
- Transferrin is also called siderophilin.
- Total iron + TIBC + Transferrin, when done together, helps in the differential diagnosis of anemia.
- The cellular uptake of iron is mediated by the cell surface transferrin receptor (TR).
- The number of transferrin receptors depends upon the cell’s need for iron.
- In the case of apoferritin deficiency, an excess of iron is deposited as small granules of Iron Oxide called hemosiderin.
What are the Transferrin functions?
- Help in the production of hemoglobin.
- Helps to regulate the amount of iron in the body.
- It binds to iron and prevents damage to the cells and tissues.
- Transferrin measurement shows the body’s ability to transport and regulate iron.
Transferrin saturation:
What sample is needed for the Transferrin saturation test?
- Take venous blood to prepare the serum.
What are the indications for the Transferrin saturation test?
- It is advised for the differential diagnosis of anemia.
- It helps in iron deficiency anemia.
- It helps in the evaluation of Thalassemia, sideroblastic anemia, and hemochromatosis.
- It differentiates hereditary hemochromatosis.
What are the precautions for the Transferrin saturation test?
- Avoid hemolysis that will interfere with the result.
- Some drugs will affect the result.
- Iron contamination of the gloves used in the test will give high values.
How will you define Transferrin saturation?
- Serum transferrin situation is obtained by dividing serum iron by TIBC.
- It is normally 20% to 50%.
- It represents the amount of iron-binding sites that are occupied.
How will you interpret Transferrin saturation?
- It is the percentage of transferrin and other iron-binding proteins.
- Transferrin saturation is calculated as follows.
- Transferrin saturation (%) = serum iron level (µg/dL) / TIBC (µg/dL) x 100
- The normal value for transferrin saturation is 20% to 50%.
- This may vary with age and sex.
- Transferrin saturation helps find the cause of abnormal iron and TIBC levels.
- Transferrin saturation is below 15% in a patient with iron deficiency anemia.
- Transferrin saturation is increased in patients:
- Hemolytic anemia.
- Sideroblastic anemia.
- Megaloblastic anemia.
- Patient with iron overload or iron poisoning.
- Hemochromatosis.
What is the clinical importance of Transferrin saturation?
- This test is crucial in evaluating abnormal iron and TIBC levels.
- It provides insights into iron metabolism and assists in diagnosing and monitoring disorders of iron homeostasis.
What is normal TIBC, Transferrin, and Transferrin saturation?
Source 1
Transferrin
| Age | mg/dL |
| 0 to 4 days | 130 to 275 |
| 3 months to 16 years | 203 to 360 |
| 16 to 60 years | |
| Male | 215 to 365 |
| Female | 250 to 380 |
| 60 to 90 years | 190 to 375 |
| >90 years | 186 to 347 |
| Maternal at term | 305 |
- To convert into SI units x 0.01 = g/L
Total Iron binding capacity (TIBC)
- Adult = 250 to 425 µg/dL
Iron saturation (%Transferrin saturation)
- Male = 20% to 50%
- Female = 15% to 50%
- To convert into SI units x 0.01 = Fraction saturation
Source 2
TIBC
- 250 to 400 µg/dL.
- It decreases in older people at around = 250 µg/dL.
- (This value varies from one reference to another)
Transferrin
- Adult = 250 to 425 mg/dL.
- Children = 203 to 360 mg/dL.
- Newborn = 130 to 275 mg /dL.
Transferrin saturation
- Male = 20% to 50 %.
- Female = 15% to 50 %.
Ferritin
- Adult male = 12 to 300 ng/mL (12 to 300 µg/L).
- Adult female = 10 to 150 ng/mL (10 to 150 µg/L).
- Children
- Newborn = 25 to 200 ng/mL.
- One-month-old = 200 to 600 ng/mL.
- 2 to 5 months old = 50 to 200 ng/mL.
- 6 months = 7 to 142 ng/mL.
What is the cause of decreased Ferritin levels?
- Iron-deficiency anemia.
What are the causes of increased Ferritin levels?
- Ferritin is an acute-phase protein raised in chronic liver diseases, alcoholism, malignancies, infection, and inflammation.
- In hemosiderosis.
- In idiopathic hemochromatosis.
- Anemias other than iron deficiency.
- Renal cell carcinoma due to hemorrhage in the tumor.
- End-stage renal diseases.
- It increases with age.
What are the causes of increased TIBC?
- Pregnancy.
- Iron deficiency.
- Acute hepatitis.
- Acute and chronic blood loss.
What are the causes of the decreased TIBC?
- Hemochromatosis.
- Hypoproteinemia in malabsorption.
- Burns.
- Cirrhosis.
- Renal diseases like nephrosis, etc.
- Thalassemia.
- Hyperthyroidism.
- Chronic diseases.
- Non-iron deficiency anemia
What are the causes of increased Transferrin?
- Iron deficiency anemia.
- Pregnancy.
- Estrogen therapy.
- Hyperestrogenism.
What are the causes of decreased Transferrin?
- Chronic infections.
- Acute inflammation.
- Microcytic anemia due to chronic diseases.
- Protein deficiency in malabsorption and burns.
- Liver disease, acute.
- Renal disease like nephrosis.
- Hemochromatosis.
- Genetic deficiency of transferrin.
What are the causes of increased Transferrin saturation?
- Hemochromatosis.
- Thalassemia.
- Hemosiderosis.
- Ingestion of iron.
- Birth control pills.
- Iron dextran injection.
What are the causes of decreased Transferrin saturation?
- Iron deficiency anemia.
- Anemia of infections and chronic diseases.
- Uremia.
- In some of the malignancies.
- Rheumatoid arthritis.
What are the findings of Differential diagnosis of various diseases?
| Disease | Total Iron | TIBC | Transferrin saturation | Ferritin |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Questions and answers:
Question 1: What is the difference between ferritin and transferrin?
Question 2: What is transferrin saturation?
- Please see more details in Total Iron.